When forming anions, these metals always lose their s-orbital electrons first. They are often good catalysts, and readily form complexes with molecules called ligands. They are usually quite dense, and are less reactive than a alkali or alkaline earth elements. They are hard solids, with high melting points and boiling points. Their compounds are often brightly colored in solution and when hydrated, and can exhibit multiple positive oxidation states. 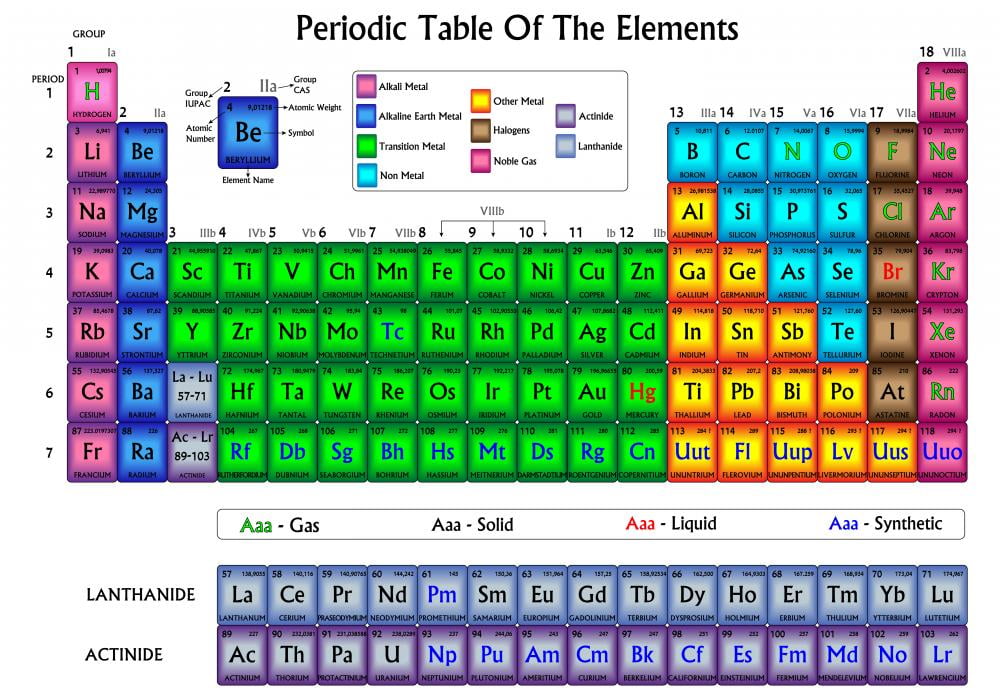
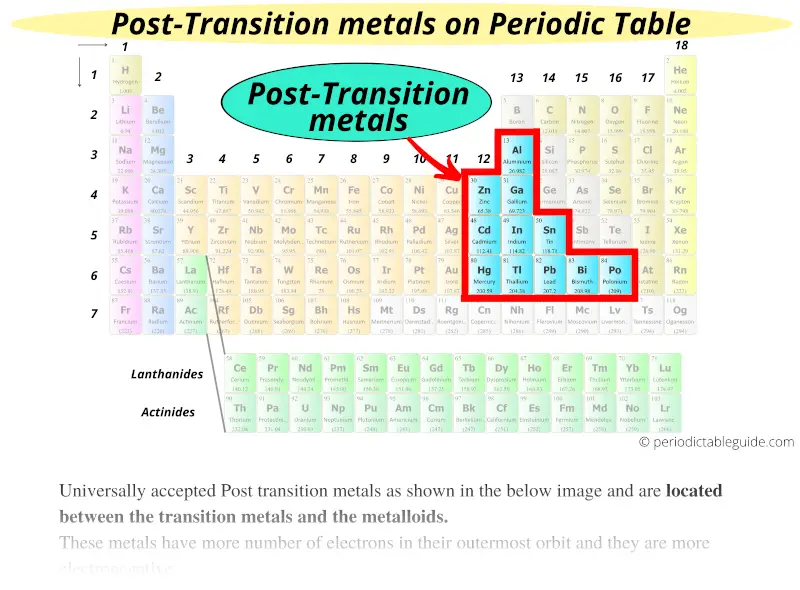
Transition metals are generally good conductors of heat and electricity, malleable and ductile. The English chemist Charles Bury first used this term, to describe this group of elements. It is sometimes used interchangeably with the term transition metal. “D block” elements is a commonly used phrase, that describes the elements in groups 3 to 12, They are called d-block elements because as you go across the rows, the d-orbital gradually gets filled with electrons. This definition excludes scandium, since the Sc+3 ion does not have unpaired d electrons. Unpaired d electrons are more likely to participate in chemical reactions. One definition of a transition metal, is any metal that has at least one unpaired d electron in one of their stable ions. There is no one universally agreed definition of what a transition metal is. Not included in this definition are the lanthanides or the actinide series elements, often called “inner transition metals”, or zinc, cadmium or mercury, which we call “post-transition metals”. 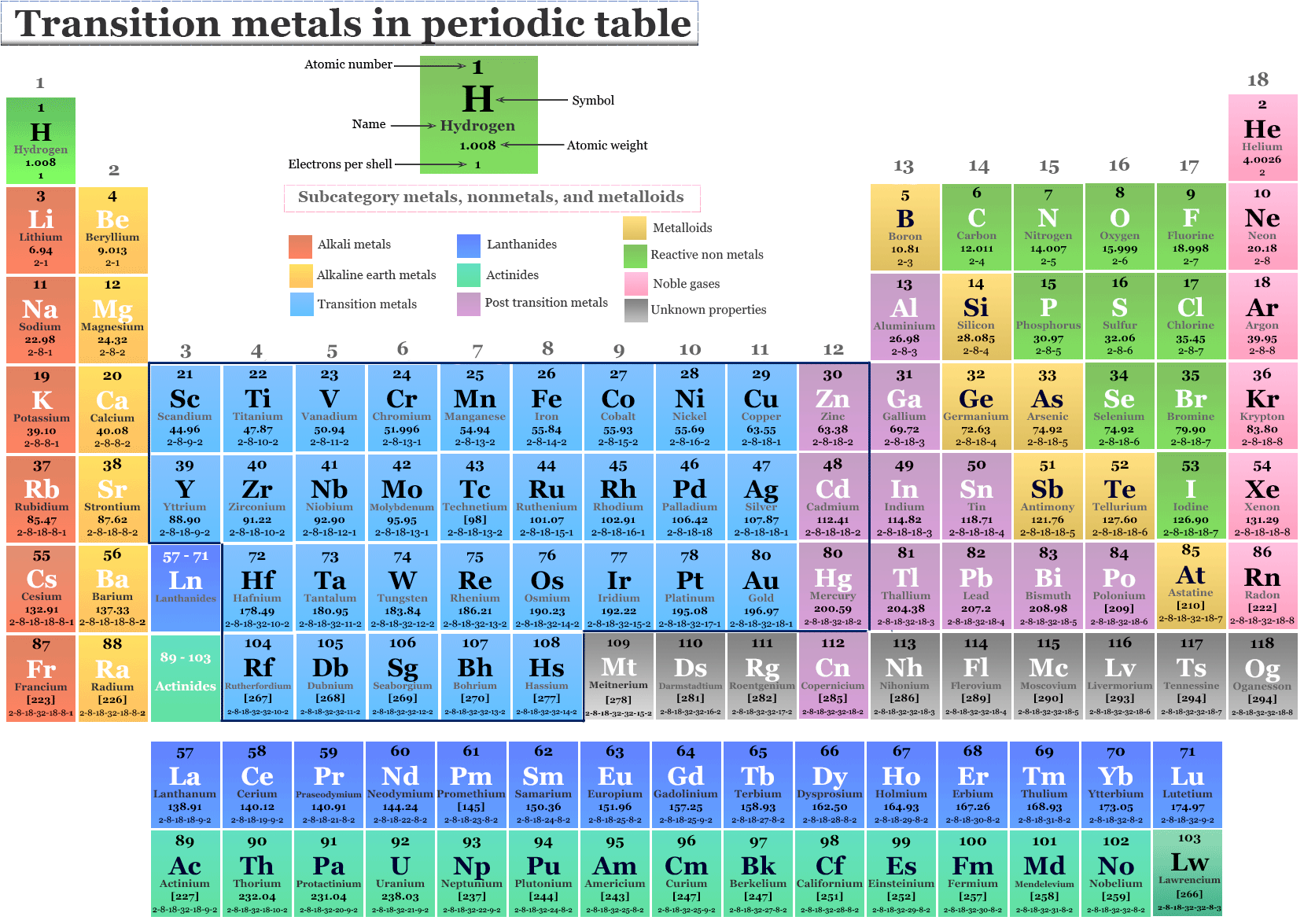
In this article, we will consider the transition metals, aka transition elements, to includes the elements of the periodic table from groups 4 to 11, plus scandium and yttrium. But what is a transition metal? There are several different definitions. When most of us think of “metals”, we are probably thinking of a transition metal.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
